University of Virginia cell biologist John Herr believes that the most ground-breaking findings always start with an insight built on basic science. He would know; his steady research on the development of sperm and eggs has led to numerous breakthroughs in reproductive health, and now he believes his team has discovered a revolutionary strategy to treat cancer.
“The focus of my work has always been to define the signature molecules that are on and within the egg and the sperm,” said Herr, a professor of cell biology in the School of Medicine. Signature molecules are those that are unique to the sperm and the egg and are not found in other tissues in the body.
The signature molecules serve as targets for tiny tracking probes that Herr and his team create using monoclonal antibodies – antibodies designed to bind to proteins originating in only one type of cell. By using the monoclonal antibodies as trackers, his team is able to detect and measure the signature molecules present. These little trackers attach to any cell carrying the signature molecules, so each monoclonal antibody can also be used as a tiny injector for targeted medication.
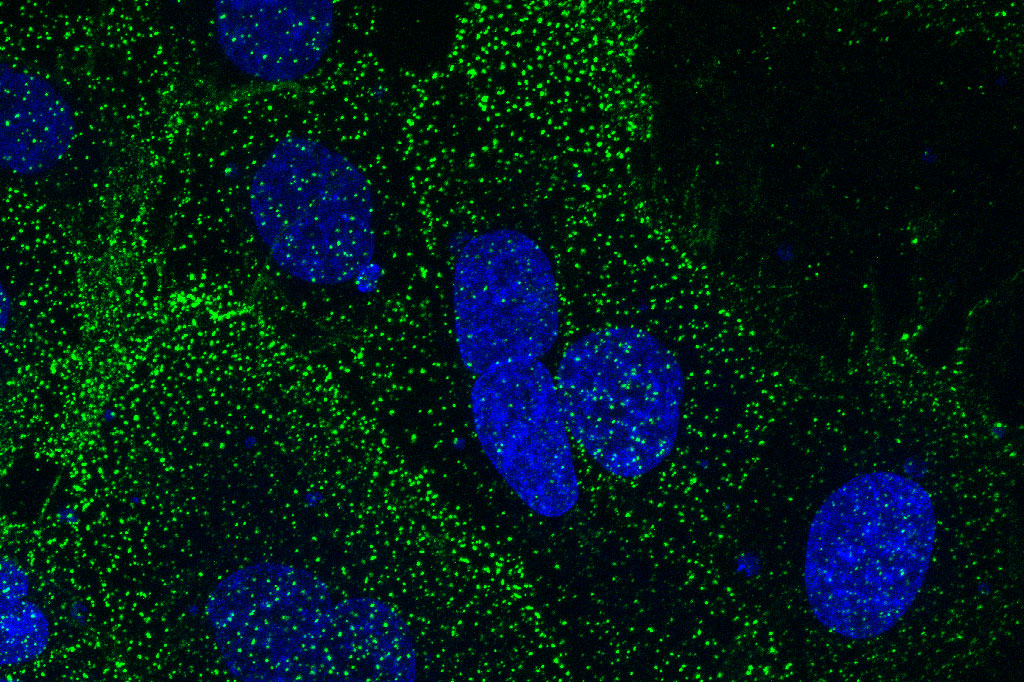
Live human uterine cancer cells, stained green where the monoclonal antibody has attached to the cell surface protein SAS1B. The blue stained structures are cell nuclei.
His oldest companies, Humagen and SpermCheck, use semen and sperm-specific proteins, respectively, to identify the presence of semen for sexual assault detection and to track sperm and determine the levels of fertile spermatozoa present. Humagen now focuses on making pipettes for manipulating the sperm and egg during in vitro fertilization and SpermCheck makes a home sperm count test.
Last year, Herr entered the female contraceptive arena with his new start-up, Ovastasis. Like his previous companies, Ovastasis focuses on protein biomarkers that are specific to the reproductive cells, in this case, the egg. The difference is that Ovastasis plans to use those protein biomarkers as targets for a novel female birth control medication.
“The identification of drug targets that are selective to the egg or to the sperm gives you opportunities to create small-molecule drugs for female and male contraception, contraceptive vaccines, and opens opportunities for biological drug strategies that selectively target the gametes in the ovary and testis,” Herr said.
Currently approved female birth control medications work by manipulating the female body’s estrogen and progesterone levels. While effective, these drugs are steroids and interact with receptors in many organ systems in the body. Unwanted side-effects of these interactions result in high discontinuation rates for the “pill.”
By using egg-specific “drug targets,” Ovastasis is developing a new form of birth control that will interact only with a woman’s reproductive cells to arrest their development. This “ovastatin” birth control method would be free of unpleasant side-effects like mood swings, acne and breast tenderness, because it would have no interaction with receptors outside the egg.
It was through the same research into egg biomarkers that Herr and his team uncovered a fundamental, and previously unknown, characteristic of cancerous tumors arising from a variety of organs.
“We discovered membrane proteins that among the normal tissues, are found only in the ovary’s population of growing eggs. These egg-specific proteins are also found in cancers that arise in a wide range of organs,” he said.
For some reason, when many cancers dysregulate, or begin to grow on their own, they revert back and take on features of a developing egg. All the body’s normal tissues differentiate from the egg during embryonic development, and acquire their own unique set of biomarkers as they begin to form specific organs.
Herr and his team are bringing awareness to an aspect of cancer that is fundamental to the disease: de-differentiation. At the same time, they’re creating an entirely new field of study into cancer-oocyte neoantigens.
“Pathologists have long appreciated that cancer cells may de-differentiate and take on histological features of embroyonic tissues. What we have now confirmed is that cancers from many organs de-differentiate to take on features of the oocyte, the original mother cell from which we all originate,” Herr said.
“We think we have a way not only to target cancer cells, but a way that could become a frontline treatment for women who have cancers of many types and want to preserve fertility.” - Dr. John Herr
With this discovery, Herr started his first cancer research company, Neoantigenics. Like Ovastasis, Neoantigenics is focused on creating a targeted drug that will affect only those cells identified by the correct cell surface biomarkers.
Their goal is to create a cancer treatment that will track and kill cells that carry the SAS1B protein, the biomarker that is found only on growing eggs and tumor cells.
The monoclonal antibodies to SAS1B can be thought of as a homing mechanism to guide a miniscule warhead selectively to the surface of cancer cells. When that warhead enters the bloodstream, it seeks out all the cells that are marked on their surfaces with SAS1B targets and then after binding on the tumor cell surface, the antibody-drug burrows inside them to release a toxic payload.
“You add an antibody with a drug on it and … within 15 minutes of contacting the cancer cells, it binds at the cell surface and begins the internalization process,” Herr said.
After about an hour, the SAS1B-marked antibodies reach compartments inside the cell and release the drug payload, triggering changes that result in cell death within a few days.
The same biomarkers that will help limit the area of impact for Ovastasis’s birth control will also help Neoantigenics confine the toxic effects of cancer treatment to growing egg and tumor cells.
This unique medication could mean a dramatic reduction of the difficult side-effects of traditional cancer treatments like hair loss, nausea, anemia and neuropathy. The drug can be used by both women and men, but for female cancer patients especially, a treatment that doesn’t touch their body’s healthy tissues is a huge breakthrough.
“We think we have a way not only to target cancer cells, but a way that could become a frontline treatment for women who have cancers of many types and want to preserve fertility,” Herr said.
While Neoantigenics’ medication would have to attack growing egg cells in order to kill similar cancer cells, it would leave a woman’s reserve of dormant primordial eggs untouched. Only egg cells that are growing in the ovary in preparation for ovulation carry the SAS1B protein. The dormant primordial eggs have not yet acquired it. After the cancer treatment is complete, those primordial eggs can begin the cycle of normal egg growth and ovulation again.
Like all of Herr’s companies, Neoantigenics was formed with the help of U.Va.’s Licensing and Venture Group, and the company will work with U.Va.’s labs as it begins testing the cancer medication – first in model organisms, and, if all goes well, they may begin human trials in select populations as early as 2016.
While he is thrilled with the implications of his discoveries, Herr’s biggest hope is that his breakthroughs demonstrate the value of investing time and public money into basic research goals. Despite his repeated success in introducing products into the commercial market, Herr has always made basic research at U.Va. his primary role.
“The general public must understand the essential link between basic science discovery research and clinical and commercial innovation,” Herr said. “Without a deep level of understanding of the fundamental biology, commercial applications are not possible. That’s what we do well as a university. That’s our task, to create a knowledge base and disseminate it into society by launching new ventures.”
Media Contact
Article Information
October 28, 2015
/content/biologist-uncovers-fundamental-new-strategy-destroying-cancer-cells

