Energy from your body heat and motion could fuel the future of preventative health care.
Researchers at the University of Virginia are developing a low-power microchip that can support on-body, real-time health monitoring. By harvesting all its needed energy from sources like body heat, motion and sunlight, the chip will provide an extremely compact, long-lasting power source for body metric sensors.
U.Va.’s chip development is part of a larger nanotechnology collaboration known as the ASSIST Center (the Nanosystems Engineering Research Center for Advanced Self-Powered Systems of Integrated Sensors and Technologies). Headquartered at North Carolina State University, the ASSIST Center’s research is funded by the National Science Foundation and includes multiple partner universities.
“The main application that we’re targeting in the center right now is childhood asthma,” said ASSIST Associate Director for Translational Research John Lach, who chairs U.Va.’s Department of Electrical and Computer Engineering.

U.Va.’s self-powering ASSIST microchip and the small circuit board where it is housed. (Photo by Sanjay Suchak / University Communications)
ASSIST is developing sensors that will work together with U.Va.’s microchip to detect changes in the body as well as environmental factors that could signal an impending asthma attack.
“We’re monitoring the person and because of that we can see the early stages of asthma exasperation and make the appropriate notifications. At the same time, we’re also monitoring the environment so we can detect what kinds of environmental exposures cause their asthma events,” Lach said. “From there, we can develop the models that allow us to intervene before an asthma event has even started.”
The data collected from asthma monitoring tests will help researchers gauge the type of demands that will be placed on their sensor-chip system, but asthma is not the only application for ASSIST technology.

Benton Calhoun (Photo by Stephanie Gross)
While some existing wearable health monitors have these capabilities, the ASSIST project is one of the first to consolidate them on a nanoscale. U.Va.’s contribution of a high-functioning, ultra-low-power microchip will allow the ASSIST team to keep their sensory devices small without the hindrances of larger batteries or communication devices.
The microchip’s most difficult task is regulating the flow of energy it takes in from the user and his or her environment. Since available body heat, motion and sunlight can fluctuate throughout the day, the chip has to adapt its function and power consumption to maintain continuous operation.
In addition to acting as a power source, the microchip also serves as a transceiver. It collects data from the attached sensors and then sends it wirelessly to another designated device, such as a user’s smartphone.
“We hope our chip will inspire a new approach to wireless sensing that leverages self-powered hardware,” Calhoun said.
The ASSIST effort is just one of the wearable technology projects that are springing up around Grounds. Here are a few other exciting devices in the works:
TEMPO

U.Va.’s TEMPO device detects and records motion, providing health care workers with more accurate data to treat elderly patients as well as those with cerebral palsy, Parkinson’s and multiple sclerosis. (Photos by Sanjay Suchak / University Communications
Health care providers are currently using it to assess the risk of falls by elderly patients.
By pinpointing balance and gait disorders, physicians and care-givers can help prevent dangerous falls before they happen. TEMPO has also been used to diagnose and treat movement difficulties for patients with cerebral palsy, Parkinson’s disease and multiple sclerosis.
BESI

The same sensor that powers TEMPO also works to detect agitated movements as part of BESI. (Photo by Sanjay Suchak / University Communications)
BESI – Behavioral and Environmental Sensing and Intervention – could allow more dementia patients to continue living with their families.
BESI combines a wearable sensor with environmental sensors placed around the homes of dementia patients.
By correlating the movements of early agitation with current environmental conditions, BESI can monitor for the first stages of agitation and identify outside factors that might be triggers. When the system detects early agitation or a common trigger, it alerts caregivers to take preventative action.
A Tumor-Fighting Skullcap

Clinical trial participant Violet Horst wears the experimental cancer-fighting device. (Photo by Jackson Smith)
Patients wear the device like a skullcap, with electrodes attached to their heads. The device is powered by a separate, portable battery attached by long wires.
U.Va.’s School of Medicine and 80 other test sites have found that 43 percent of newly diagnosed patients who wear the device survived two years following diagnosis.
Only 29 percent of those who didn’t wear the device lived that long.
The Artificial Pancreas
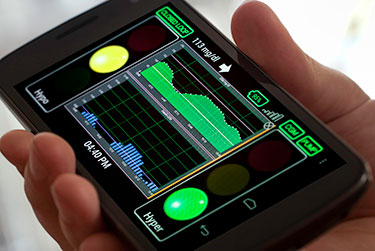
The Artificial Pancreas is an adapted smartphone that incorporates a glucose monitor and automatic insulin pump. It offers patients with type-1 diabetes a rare relief from the fear of dangerous low blood sugar episodes.
For people with type-1 diabetes, the goal of the artificial pancreas is to alleviate the fear of life-threatening low blood sugar episodes.
The artificial pancreas technology – known as the Diabetes Assistant– is designed to automatically monitor and regulate blood-sugar levels using a reconfigured smart phone running advanced algorithms and linked wirelessly to a continuous glucose monitor and an insulin pump.
Media Contact
Article Information
June 15, 2015
/content/new-microchip-improves-future-self-powered-wearable-technology

